On the origin of high ionic conductivity in Na-doped SrSiO3†
Abstract
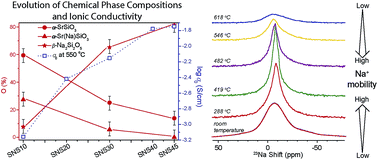
Understanding the local structure and ion dynamics is at the heart of ion conductor research. This paper reports on high-resolution solid-state 29Si, 23Na, and 17O NMR investigation of the structure, chemical composition, and ion dynamics of a newly discovered fast ion conductor, Na-doped SrSiO3, which exhibited a much higher ionic conductivity than most of current oxide ion conductors. Quantitative analyses reveal that with a small dose (<10 mol%) of Na, the doped Na integrates into the SrSiO3 structure to form NaxSr1−xSiO3−0.5x, and with >10 mol% Na doping, phase separation occurs, leading to the formation of an amorphous phase β-Na2Si2O5 and a crystalline Sr-rich phase. Variable-temperature 23Na and 17O magic-angle-spinning NMR up to 618 °C have shown significant changes in Na ion dynamics at high temperatures but little oxide ion motion, suggesting that Na ions are responsible for the observed high ionic conductivity. In addition, β-Na2Si2O5 starts to crystallize at temperatures higher than 480 °C with prolonged heating, resulting in reduction in Na+ motion, and thus degradation of ionic conductivity. This study has contributed critical evidence to the understanding of ionic conduction in Na-doped SrSiO3 and demonstrated that multinuclear high-resolution and high-temperature solid-state NMR is a uniquely useful tool for investigating ion conductors at their operating conditions.


 Please wait while we load your content...
Please wait while we load your content...