Successive C–C bond cleavage, fluorination, trifluoromethylthio- and pentafluorophenylthiolation under metal-free conditions to provide compounds with dual fluoro-functionalization†
Abstract
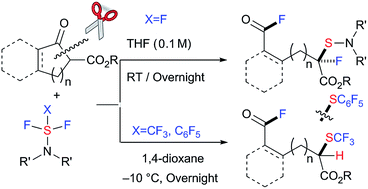
The selective C–C bond cleavage and simultaneous formation of two C–F bonds and one C–S bond in β-keto esters with nucleophilic fluorination reagents such as DAST under metal-free/catalyst-free conditions is disclosed. Double fluorination at two remote carbons with additional dialkylamino-sulfenylation provided unique fluorinated compounds in good to high yields. This method can be applied for the successive C–C bond cleavage/fluorination/trifluoromethylthiolation of β-keto esters using trifluoromethyl-DAST (CF3-DAST) providing different type of fluorinated and trifluoromethylthiolated compounds via a shunt pathway. Doubly fluoro-functionalized compounds obtained in these reactions are unique and difficult to synthesize by other methods.

 Please wait while we load your content...
Please wait while we load your content...