Encapsulation of an organometallic cationic catalyst by direct exchange into an anionic MOF†
Abstract
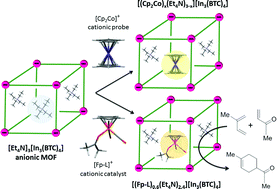
Metal–Organic Frameworks (MOFs) are porous crystalline materials that have emerged as promising hosts for the heterogenization of homogeneous organometallic catalysts, forming hybrid materials which combine the benefits of both classes of catalysts. Herein, we report the encapsulation of the organometallic cationic Lewis acidic catalyst [CpFe(CO)2(L)]+ ([Fp–L]+, Cp = η5-C5H5, L = weakly bound solvent) inside the pores of the anionic [Et4N]3[In3(BTC)4] MOF (H3BTC = benzenetricarboxylic acid) via a direct one-step cation exchange process. To conclusively validate this methodology, initially [Cp2Co]+ was used as an inert spatial probe to (i) test the stability of the selected host; (ii) monitor the stoichiometry of the cation exchange process and (iii) assess pore dimensions, spatial location of the cationic species and guest-accessible space by single crystal X-ray crystallography. Subsequently, the quasi-isosteric [Fp–L]+ was encapsulated inside the pores via partial cation exchange to form [(Fp–L)0.6(Et4N)2.4][In3(BTC)4]. The latter was rigorously characterized and benchmarked as a heterogeneous catalyst in a simple Diels–Alder reaction, thus verifying the integrity and reactivity of the encapsulated molecular catalyst. These results provide a platform for the development of heterogeneous catalysts with chemically and spatially well-defined catalytic sites by direct exchange of cationic catalysts into anionic MOFs.

 Please wait while we load your content...
Please wait while we load your content...