K2xSn4−xS8−x (x = 0.65–1): a new metal sulfide for rapid and selective removal of Cs+, Sr2+ and UO22+ ions†
Abstract
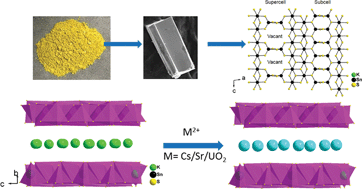
The fission of uranium produces radionuclides, 137Cs and 90Sr, which are major constituents of spent nuclear fuel. The half-life of 137Cs and 90Sr is nearly 30 years and thus that makes them harmful to human life and the environment. The selective removal of these radionuclides in the presence of high salt concentrations from industrial nuclear waste is necessary for safe storage. Here we report the synthesis and crystal structure of K2xSn4−xS8−x (x = 0.65–1, KTS-3) a material which exhibits excellent Cs+, Sr2+ and UO22+ ion exchange properties in varying conditions. The compound adopts a layered structure which consists of exchangeable potassium ions sandwiched between infinite layers of octahedral and tetrahedral tin centers. K2xSn4−xS8−x (x = 0.65–1, KTS-3) crystallizes in the monoclinic space group P21/c with cell parameters a = 13.092(3) Å, b = 16.882(2) Å, c = 7.375(1) Å and β = 98.10(1)°. Refinement of the single crystal diffraction data revealed the presence of Sn vacancies in the tetrahedra that are long range ordered. The interlayer potassium ions of KTS-3 can be exchanged for Cs+, Sr2+ and UO22+. KTS-3 exhibits rapid and efficient ion exchange behavior in a broad pH range. The distribution coefficients (Kd) for KTS-3 are high for Cs+ (5.5 × 104), Sr2+ (3.9 × 105) and UO22+ (2.7 × 104) at neutral pH (7.4, 6.9, 5.7 ppm Cs+, Sr2+ and UO22+, respectively; V/m ∼ 1000 mL g−1). KTS-3 exhibits impressive Cs+, Sr2+ and UO22+ ion exchange properties in high salt concentration and over a broad pH range, which coupled with the low cost, environmentally friendly nature and facile synthesis underscores its potential in treating nuclear waste.

 Please wait while we load your content...
Please wait while we load your content...