Electrosynthesis and characterization of nanocrystalline UO2 coating from aqueous alkaline electrolyte†
Abstract
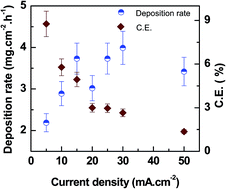
In this study, UO2 coating was deposited by direct current (DC) electrolysis from an aqueous electrolyte containing UO2(NO3)2 as source of uranium. Deposition parameters such as electrolyte pH, temperature and current density were optimized in order to obtain a smooth adherent UO2 coating on metallic substrates. The effect of pH, temperature and current density on the deposition rate and on faradic current efficiency (CE) was studied in detail. The study revealed that the control over pH of the electrolyte was more important in comparison to temperature and current density to obtain a smooth deposition of UO2 from this uranyl nitrate–oxalate complex electrolyte. Grazing incidence X-ray diffraction (GI-XRD) of the as-deposited coating with a broad peak confirmed amorphous to ultra-nanocrystalline deposition. Upon vacuum annealing at higher temperature, the as-deposited coating transformed into nanocrystalline fcc UO2 with the appearance of sharper (111), (220), (220), (222) XRD reflections. The average grain size obtained was 13.2 nm for the sample annealed at 700 °C. In the presence of air, the fcc UO2 coating was transformed largely into the hcp U3O8 phase with a small percentage of hcp UO3 at temperatures higher than 400 °C. The surface morphology of the as-deposited coating showed a granular morphology with the appearance of few cracks. Upon annealing at 700 °C, the film showed a network of cracks throughout the surface with numerous pores inside the UO2 matrix. Composition analysis by EDS of the film confirmed the presence of uranium and oxygen. Cross-sectional FESEM analysis of the focused ion beam (FIB) cut samples confirmed deposition of a 6 μm thick UO2 coating. Upon annealing at 700 °C, it converted into a highly porous UO2 coating with improved adhesion with the substrate.

 Please wait while we load your content...
Please wait while we load your content...