Solvent effects on adsorption kinetics, dye monolayer, and cell performance of porphyrin-sensitized solar cells†
Abstract
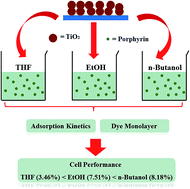
The effect of three dye loading solvents (THF, ethanol, and n-butanol) on the adsorption kinetics, the binding mode of the dye molecules on the TiO2 nanoparticles, and photovoltaic performance in dye-sensitized solar cells was reported. The kinetic study indicated that solvents greatly affected the adsorption amount and adsorption rate on the surface of TiO2 nanoparticles. The adsorption data in THF, ethanol and n-butanol were analyzed by applying an intraparticle diffusion model, pseudo-first order model, and pseudo-second order model, respectively. It was found that adsorption rate was controlled by mass transfer processes. UV-vis spectra and ATR-FTIR spectra of porphyrin-coated electrodes were measured to study the intermolecular interaction and binding mode on TiO2 films, respectively. Finally, the cell performance was tested under different solvents and the best efficiency of 8.18% was obtained when n-butanol was used as the solvent for dye-loading.


 Please wait while we load your content...
Please wait while we load your content...