Efficient removal of HCN through catalytic hydrolysis and oxidation on Cu/CoSPc/Ce metal-modified activated carbon under low oxygen conditions
Abstract
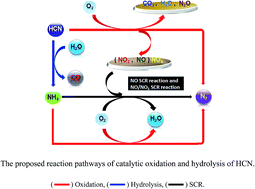
The hydrogen cyanide (HCN) removal efficiency of activated carbon modified with different metal was studied under low oxygen conditions. When activated carbon was modified with Cu(NO)3, cobalt sulfonated phthalocyanine (CoSPc) and Ce(NO3)3·6H2O, its catalytic efficiency in HCN removal was significantly enhanced with the optimal conditions of 400 °C as the calcination temperature, 10% relative humidity and 1% oxygen concentration. And the catalytic hydrolysis and oxidation efficiency of AC–Cu–CoSPc–Ce was more than 98% at 200–350 °C with the maximum selectivity to N2 of 52.6% at 300 °C. Although both AC–Cu and AC–Cu–CoSPc–Ce catalysts were reliable and stable, AC–Cu–CoSPc–Ce had a better catalytic activity at lower temperature. According to BET and X-ray photoelectron spectroscopy (XPS) results, Cu was mainly in the form of CuO and Cu2O, Co was present in the form of Co2+ and Co0, and Ce was mainly in the form of CeO2 and Ce2O3 on the catalyst surface. The reaction pathways were proposed.

 Please wait while we load your content...
Please wait while we load your content...