Palladium(ii)-catalyzed hydroxy-involved enolate-type efficient C–C functionalization of hydroxynaphthoquinones at room temperature†
Abstract
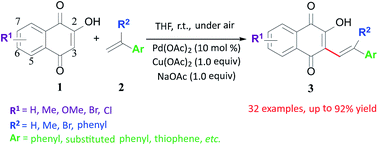
A simple, mild, efficient (46–92%) and scalable strategy for the synthesis of hydroxynaphthoquinones was developed through a Pd-catalyzed hydroxy-involved enolate-type C–C bond formation reaction at room temperature. The functional group tolerance exhibited by this reaction, along with the possibility of scalable production, make it a method of choice for synthesizing diverse hydroxynaphthoquinones.


 Please wait while we load your content...
Please wait while we load your content...