An efficacy of ‘nano’ in brannerite-type CoV2O6 conversion electrode for lithium batteries
Abstract
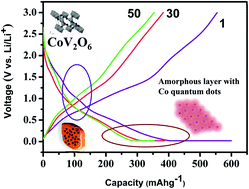
Among the classical anodes, conversion anodes play a unique role due to their capability to provide higher initial discharge capacity than the theoretical capacity. Poor capacity retention and voltage hysteresis exhibited by classical conversion anodes are the major obstacles for their commercialization in lithium battery market. Nanotechnology may be a suitable contrivance to tackle this problem. To our knowledge, this is the first attempt to synthesize brannerite-type nano sized cobalt vanadate by rheological phase reaction method and use it as a conversion anode in lithium batteries. Although this material provides a high initial irreversible capacity of around 600 mA h g−1 in its preliminary cycle, stabilization of capacity with approximately 100% coulombic efficiency and considerable low voltage hysteresis in the proceeding cycles may place this material in the limelight for its development as a commercially available anode material. The electrochemical conversion of CoO nanoparticles into Co quantum dots occurred in an amorphous lithiated vanadium oxide matrix, which functioned as a source of reactive sites, as well as a separator to preserve the nanoparticles against further agglomeration, thereby, offering a better rate capability.

 Please wait while we load your content...
Please wait while we load your content...