High pressure studies of trimethyltin azide by Raman scattering, IR absorption, and synchrotron X-ray diffraction
Abstract
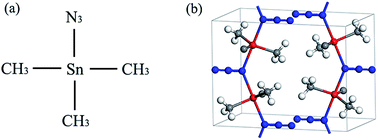
We present the high-pressure behavior of energetic material trimethyltin azide ((CH3)3SnN3, TMSnA) by in situ Raman scattering, IR absorption, and synchrotron angle-dispersive X-ray diffraction techniques with pressure up to 35 GPa at room temperature. Raman and IR spectra analyses provide evidence for two reversible phase transitions at 1.4 and 6.6 GPa, respectively, which were confirmed by XRD results. The first phase transition at 1.4 GPa is attributed to the rotation of CH3 group, resulting in the symmetry transformation from Pnna to P222. Upon compression, the distortion of the unit cell induced a significant modification of the CH3 group leading to the second phase transition at 6.6 GPa. Further analyses of the Raman and IR spectra reveal that the azide group becomes increasingly asymmetric with increasing pressure. At 35.2 GPa, all the Raman vibration modes of the azide group vanish, which means the molecular azide group transforming into a nonmolecular state. The previous studies indicate that the polymerization of nitrogen undergoes an amorphization process. We thus anticipate the high-pressure study of TMSnA which may offer a promising strategy for exploiting and improving in the formation of polymeric nitrogen of energetic azides.

 Please wait while we load your content...
Please wait while we load your content...