Visible light driven efficient N and Cu co-doped ZnO for photoinactivation of Escherichia coli
Abstract
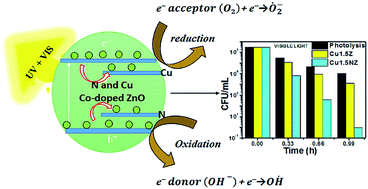
Co-doping of a transition metal ion (Cu) and non-metal ion (N) on metal oxide substrate ZnO was achieved for the first time. The transition metal doping and N doping was carried out using combustion synthesis and a hydrothermal method, respectively. The combined effect of Cu and N doping was observed by varying the Cu percentage and keeping the nitrogen doping percentage constant. The catalysts were characterized by a wide variety of techniques. Incorporation of Cu and N in the ZnO matrix introduces sub energy bands near the conduction band and valence band, respectively and facilitates the absorption of longer wavelength spectra. These sub energy levels can also act as trapping sites for excitons. This results in reduced recombination and enhanced photocatalytic activity as confirmed by photoluminescence (PL). The photocatalytic inactivation of Gram negative bacteria (E. coli) was investigated using the N and Cu co-doped ZnO under UV and visible irradiation with different atom% of Cu. Kinetic analysis was used to obtain the order and rate constant of the inactivation reactions.


 Please wait while we load your content...
Please wait while we load your content...