Hematite decorated multi-walled carbon nanotubes (α-Fe2O3/MWCNTs) as sorbents for Cu(ii) and Cr(vi): comparison of hybrid sorbent performance to its nanomaterial building blocks†
Abstract
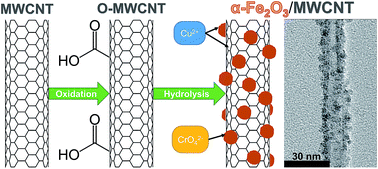
Major hurdles in the application of engineered nanomaterials for water treatment include loss of reactive surface area arising from particle aggregation and the development of application platforms that limit their potential release into the treated water supply. Here, we develop hybrid nanostructures through the growth of hematite (α-Fe2O3) nanoparticles, which are recognized sorbents for various heavy metals, on multi-walled carbon nanotubes (MWCNTs). The hybrid nanostructures were synthesized via hydrolysis of ferric nitrate in the presence of carboxylated MWCNTs, and their activity as sorbents toward Cu(II) and chromate (CrO42−) was examined as a function of pH (i.e., pH-edge experiments) and initial metal concentration (i.e., adsorption isotherms). Characterization of α-Fe2O3/MWCNT nanostructures via Raman spectroscopy and transmission electron microscopy (TEM) with selected area electron diffraction (SAED) confirmed the deposited iron phase as α-Fe2O3. Further, complementary acid digestions and TEM imaging revealed that the amount (0.1 and 0.5 g g−1 α-Fe2O3/MWCNT) and size [5.9 (±1.1) and 8.9 (±1.5) nm, respectively] of α-Fe2O3 nanoparticles immobilized on MWCNTs were tunable during synthesis. Generally, mass-normalized concentrations of adsorbed Cu(II) and CrO42− were greatest for α-Fe2O3/MWCNT hybrids relative to adsorption on either carboxylated MWCNTs or freely suspended α-Fe2O3 nanoparticles, with evidence implicating α-Fe2O3 as the active sorbent phase in hybrid materials. Indeed, per unit mass of available α-Fe2O3, hybrid sorbents exhibited capacities comparable to or exceeding most other iron-based sorbents for Cu(II) and CrO42− (from 220 to 470 mg Cu(II) per g α-Fe2O3 and 60 mg CrO42− per g α-Fe2O3, respectively, at pH 6 and 20 °C). The enhanced sorption capacity of the hybrid nanostructures is due, at least in part, to the greater available surface area of α-Fe2O3 nanoparticles immobilized on MWCNTs when compared to their more extensively aggregated state in suspension. Notable differences in the pH-dependent trends of Cu(II) and CrO42− uptake on α-Fe2O3/MWCNT hybrids, along with differences in zeta potential measurements across pH, also suggest that the immobilized α-Fe2O3 nanoparticles may exhibit unique surface reactivity relative to their freely suspended analogs as a result of their association with the negatively charged MWCNT surface.

 Please wait while we load your content...
Please wait while we load your content...