Enhanced adsorption of Cu(ii) ions on chitosan microspheres functionalized with polyethylenimine-conjugated poly(glycidyl methacrylate) brushes†
Abstract
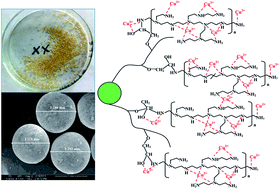
To enhance the adsorption capacity of crosslinked chitosan (CCS) microspheres towards Cu(II) ions, well-defined poly(glycidyl methacrylate) (PGMA) brushes were grafted onto the surfaces of CCS microspheres via surface-initiated atom transfer radical polymerization (ATRP) for subsequent conjugation of polyethylenimine (PEI). The resultant PEI-grafted CCS (defined as CCS-g-PGMA-c-PEI) microsphere was used as an effective adsorbent to uptake Cu(II) ions from aqueous solution. Success in each functionalization step was ascertained by ATR-FTIR, XPS, SEM and water contact angle measurements. Batch adsorption experiments were performed to determine adsorption kinetics, adsorption isotherms and thermodynamics of Cu(II) ions on the CCS-g-PGMA-c-PEI microspheres. The adsorption equilibrium of Cu(II) ions on the microspheres was found to be rapidly established within 60 min at an optimal solution pH of 5.0, and the adsorption kinetics was well represented by the pseudo-second-order model, together with a significant effect of mass transfer on the Cu(II) adsorption rate. The Langmuir-fitted maximum adsorption capacity was about 3.58 mmol g−1 (229 mg g−1), and the calculated thermodynamic parameters demonstrated an endothermic and spontaneous adsorption process of Cu(II) ions. The PEI-grafted CCS microspheres also exhibited good regenerability and stability for recycle applications. The postulated adsorption mechanism was proposed to account for the adsorption process of Cu(II) ions on the CCS-g-PGMA-c-PEI microspheres.

 Please wait while we load your content...
Please wait while we load your content...