Fabrication and covalent modification of highly chelated hybrid material based on silica-bipyridine framework for efficient adsorption of heavy metals: isotherms, kinetics and thermodynamics studies
Abstract
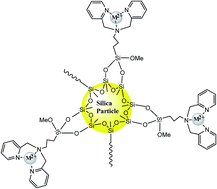
Adsorbent materials are essential in clean-up processes. Research of efficient materials is a well-established technology. In this work, a novel and excellent host for heavy metals was synthesized by covalent immobilization of bipyridine tripodal receptor onto silica particles. The new engineered surface was well analyzed and evaluated by BET, BJH, EA, FT-IR, SEM, TGA and solid-state 13C NMR. The adsorption properties were investigated using Pb(II), Cd(II), Zn(II) and Cu(II) metals by varying all relevant parameters such as pH, contact time, concentration, thermodynamic parameters, kinetics, Langmuir and Freundlich isotherms, etc. The hybrid material has been found to exhibit high distribution coefficients for heavy metals. Adsorption kinetics follows a pseudo-second-order model, as a rapid process as evidenced by equilibrium achieved within 20 min. The resulting adsorption isotherms of the material were better represented by the Langmuir model than the Freundlich model. The thermodynamic parameters (ΔH°, ΔS° and ΔG°) revealed that the adsorption was endothermic and spontaneous. In addition, the proposed material demonstrates a high degree of reusability over a number of cycles, thus enhancing its potential for application in heavy metals recycling. All metal ions were determined by atomic absorption measurements.


 Please wait while we load your content...
Please wait while we load your content...