From mononuclear to linear one-dimensional coordination species of copper(ii)–chloranilate: design and characterization†
Abstract
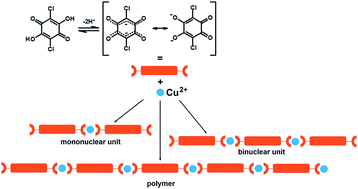
A series of six novel mononuclear, binuclear and linear one-dimensional (1D) compounds of copper(II) with chloranilic acid (3,6-dichloro-2,5-dihydroxybenzoquinone, H2CA) is prepared and a design strategy for the preparation of such complexes is discussed. Four described compounds are linear 1D coordination polymers [Cu(CA)]n, whereas another two involve a binuclear and a mononuclear, Cu2(CA)3 and Cu(CA)2, core unit. A linear polymer incorporating bulky aromatic imidazole has been synthesized as a result of investigation of the influence of pH on the reaction mixture. Two coordination modes of the chloranilate dianion are observed. The bridging (bis)bidentate mode generates linear 1D polymeric species. Among these one reveals square-pyramidal coordination of Cu2+, whereas the three polymers contain Cu2+ in an octahedral arrangement. However, the combination of both, terminal bidentate (ortho-quinone) and bridging (bis)bidentate modes of coordination produces a binuclear complex anion, which comprises a square-pyramidal coordination of Cu2+ complex anions forming a supramolecular honeycomb-like network encapsulating 4,4′-bipyridine cations. When the chloranilate dianion coordinates the Cu2+ atom only in a terminal bidentate mode, a mononuclear complex with an octahedral environment of the metal centre is formed. The presence of the bulky ancillary ligand imidazole produces an unprecedented packing involving chiral (racemic) and achiral (meso-compound) coordination polymers in the same crystal. Electron spin resonance spectroscopy of polycrystalline samples determined g-tensor parameters of copper(II) ions in different coordination geometries and revealed weak exchange interactions (|J| < 1 cm−1) in linear metal-complex polymers and dimeric species.

 Please wait while we load your content...
Please wait while we load your content...