An efficient non-reaction based colorimetric and fluorescent probe for the highly selective discrimination of Pd0 and Pd2+ in aqueous media†
Abstract
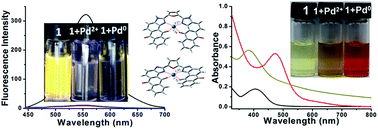
A novel anthraquinone-imidazole based colorimetric and fluorogenic probe 1 is synthesized, which can discriminate the oxidation states of palladium (Pd0 and Pd2+) by naked eye with high selectivity in aqueous media owing to the difference in coordination within the right sized pocket of the probe molecule. The experimental results (fluorescence, UV/Vis and 1H NMR spectroscopy) aided by density functional theory (DFT) calculations reveal that charge transfer (CT) is the main cause of the selective detection of the oxidation states of palladium. In the 1–Pd2+ complex, which is deficient in electrons, all the electron transitions represent electron transfers from the probe molecule to the central metal atom i.e. partial ligand-to-metal transition excitation while electronically filled Pd0 shows electron transfers from the metal atom to the probe ligand i.e. partial metal-to-ligand transition excitation.

 Please wait while we load your content...
Please wait while we load your content...