Understanding the structural changes and depolymerization of Eucalyptus lignin under mild conditions in aqueous AlCl3†
Abstract
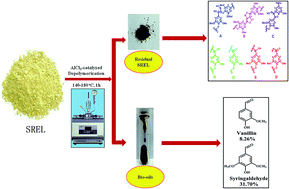
Lignin is a unique renewable source of phenolic products for the potential replacement of fossil fuels. In this study, swollen residual enzyme lignin (SREL) was prepared and then depolymerized in aqueous AlCl3 to produce phenolic compounds. The residual lignin after depolymerization was further characterized by FT-IR, advanced NMR (2D-HSQC, 13C, and 31P NMR spectra) and GPC techniques, and the degradation products were identified and quantified by GC-MS. The results showed that the depolymerization reaction became the leading reaction under these conditions. Meanwhile, the depolymerization process also resulted in a large amount of a volatile fraction, which was composed of phenols, guaiacols, and syringols. Among the volatile fraction, syringaldehyde accounted for about 31.70% of the bio-oil under optimum conditions (at 160 °C for 1 h). Furthermore, the mechanism for lignin depolymerization in aqueous AlCl3 was discussed and an enhanced understanding of the effects of aqueous AlCl3 pretreatment on lignin chemistry was provided, which will improve the pretreatment methodology to reduce biomass recalcitrance.

 Please wait while we load your content...
Please wait while we load your content...