Diethyldithiocarbamate (DDTC) induced formation of positively charged silver nanoparticles for rapid in situ identification of inorganic explosives by surface enhanced Raman spectroscopy†
Abstract
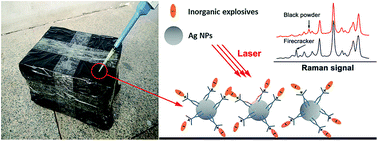
The nonvolatile inorganic explosives deposition on various surfaces requires rapid in situ detection by homeland security. Surface enhanced Raman spectroscopy (SERS), a prompt and sensitive detection method, has been widely applied in chemical and biological samples analysis. The most popular noble metal nanoparticle colloids, prepared by chemical reduction with citrate or hydroxylamine, are generally negatively charged. This study demonstrates that the negatively charged silver nanoparticles were transformed into the positively charged ones by sodium diethyldithiocarbamate (DDTC), a potent copper chelating agent. The zeta potential of DDTC modified silver colloid is about 32.1 mV, suggesting its positively charged nature. The XPS and SERS spectra indicate the positively charged silver nanoparticles resulted from the bidentate configuration leading to the redistribution of the free electron pair in N atom and formation of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+ bonding of DDTC. The positively charged silver nanoparticles can be used in the detection of inorganic explosives anions with a subnanogram detection limit through electrostatic interactions. The common organic explosive picric acid (PA) could also be detected through a similar interaction force.
N+ bonding of DDTC. The positively charged silver nanoparticles can be used in the detection of inorganic explosives anions with a subnanogram detection limit through electrostatic interactions. The common organic explosive picric acid (PA) could also be detected through a similar interaction force.

 Please wait while we load your content...
Please wait while we load your content...