Electrochemical properties of gold and glassy carbon electrodes electrografted with an anthraquinone diazonium compound using the rotating disc electrode method†
Abstract
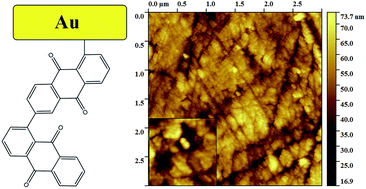
In this paper, gold and glassy carbon (GC) electrodes were electrochemically grafted with anthraquinone (AQ) groups derived from Fast Red AL salt via electrochemical reduction of AQ diazonium cations. For the first time, the electrografting was performed by combining the cyclic voltammetry (CV) or potential step method with the rotating disc electrode (RDE) technique in order to prepare thick films of AQ on Au and GC electrodes. For comparison purposes, a CV or potential step method without the RDE was also used. The attachment of thick AQ films to the Au electrode surface was confirmed by X-ray photoelectron spectroscopy and atomic force microscopy. The highest surface concentration value (ΓAQ) of AQ on the Au and GC electrodes (ca. 2.1 × 10−8 and 1.8 × 10−8 mol cm−2, respectively) was obtained by employing the combination of CV and RDE methods during the electrografting procedure. The oxygen reduction reaction results revealed that the electrochemical behaviour of AQ-modified Au electrodes does not depend on the ΓAQ value similar to the GC electrodes modified with thick AQ films. However, some differences were observed when the blocking effect of thick AQ films on Au and GC electrodes towards the ferri/ferrocyanide redox probe was studied.

 Please wait while we load your content...
Please wait while we load your content...