Exploration of the diastereoselectivity in an unusual Grignard reaction and its application towards the synthesis of styryl lactones 7-epi-(+)-goniodiol and 8-epi-(−)-goniodiol†
Abstract
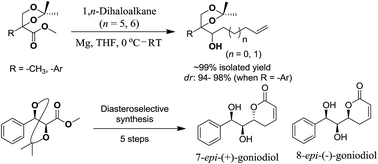
An unusual diastereoselective Grignard reaction is explored, where the Grignard reagents are derived from 1,n-dihaloalkanes. A steric bias due to the presence of a quaternary centre adjacent to the acetonide ester at the benzylic position is responsible for the formation of an intramolecularly reduced product in almost quantitative yield. This steric hindrance is responsible for the diastereoselectivity observed with a variety of aromatic as well as aliphatic esters. The unusual Grignard reaction furnishes long chain secondary alcohols possessing a terminal olefin, which are synthetically important intermediates. As an application of this method, the diastereoselective synthesis of styryl lactones viz. 7-epi-(+)-goniodiol (29) and 8-epi-(−)-goniodiol (30) has been achieved.

 Please wait while we load your content...
Please wait while we load your content...