Multi-substituted triazatruxene-functionalized pyrene derivatives as efficient organic laser gain media†
Abstract
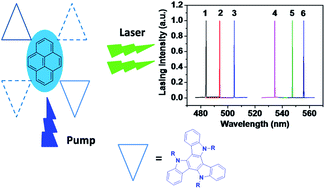
A set of pyrene derivatives substituted with multiple triazatruxene at the 1, 6- and 1, 3, 6, 8-positions, namely Py-2TAT and Py-4TAT, was synthesized and characterized. Their thermal, photophysical, and electrochemical properties were investigated in comparison with those of the pyrene and triazatruxene units to explore the relationship between the molecular architectures and corresponding properties. It is found that introducing a triazatruxene unit onto the rigid pyrene chromophore can effectively depress the crystalline nature of the pyrene and triazatruxene units, which endow the resulting materials with improved morphology properties, enhanced thermal stabilities, and favorable solution processiblity. Particularly, due to the integration of triazatruxene functional units, the resulting materials exhibit high-lying HOMO energy levels that are well matched with that of the PEDOT:PSS/ITO anode, leading to an improved hole-injection property. Solution-processed non-doped films exhibited prominent amplified spontaneous emission (ASE) and lasing characteristics. Low ASE thresholds of 150 nJ per pulse (1.4 kW cm−2) at 508 nm for Py-2TAT and 450 nJ per pulse (4.1 kW cm−2) at 530 nm for Py-4TAT were recorded. Consequently, one-dimensional (1D) distributed feed-back (DFB) lasers with low lasing threshold of 21 nJ per pulse (8.6 kW cm−2) was demonstrated. The selection of different combinations of grating periods and film thicknesses has provided the opportunity to fine tune the lasing wavelength of the DFB lasers in the range of 484–556 nm. The results indicated that construction of multi-substituted triazatruxene-pyrene architectures was beneficial to improving the electrical properties and thermal stabilities without largely sacrificing the optical properties, manifesting the potential of triazatruxene-functionalized pyrenes as efficient gain media for electrically pumped organic lasers.

 Please wait while we load your content...
Please wait while we load your content...