Exploration of the electrochemical mechanism of ultrasmall multiple phases molybdenum carbides nanocrystals for hydrogen evolution reaction†
Abstract
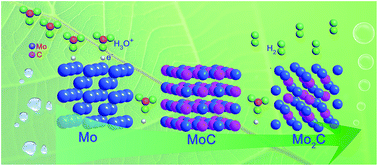
Sustainable and affordable hydrogen production through splitting of water, an essential step towards renewable and clean energy storage, calls for efficient non-precious-metal catalysts to make the process economically viable. Ultrasmall multiple phases molybdenum carbides nanocrystals (2.5 nm for MoC and 5.0 nm for Mo2C) on graphene support were synthesized by a simple in situ method. Both molybdenum carbides on graphene hybrid materials, the MoC-G and Mo2C-G, show extraordinary high activity for hydrogen evolution reaction (HER) in acid media. The reaction kinetics of the MoC-G and Mo2C-G were revealed. The X-ray photoelectron spectroscopy (XPS) and X-ray absorption fine structure (XAFS) were conducted to study the electronic nature of MoC-G and Mo2C-G electrocatalysts to explore the electrochemical mechanism of ultrasmall multiple phases molybdenum carbides nanocrystals on graphene for hydrogen evolution reaction.
- This article is part of the themed collection: Nanoscience and nanotechnology in electrochemistry

 Please wait while we load your content...
Please wait while we load your content...