Catalytic dehydration of 2,3-butanediol over P/HZSM-5: effect of catalyst, reaction temperature and reactant configuration on rearrangement products
Abstract
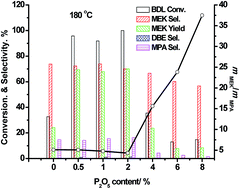
As a type of important bio-based vicinal diol, 2,3-butanediol could be transformed into methyl ethyl ketone and 2-methyl propanal through a pinacol rearrangement mechanism under acid catalysis conditions. In this paper, a series of P/HZSM-5 (Si/Al = 360) samples with various phosphate contents were prepared and tested via the catalytic transformation of 2,3-butanediol, with particular focus on the effect of phosphate content on the ratio of methyl ethyl ketone to 2-methyl propanal. The catalyst structures were studied using several physico-chemical methods such as XRD, N2 sorption, NH3-TPD and FT-IR. At 180 °C, the ratio of methyl ethyl ketone to 2-methyl propanal increased from 5.1 to 37.5 when the content of phosphate increased from 0.5 to 8.0. When the reaction temperature increased from 180 °C to 300 °C over 4% P2O5/HZSM-5, the ratio of methyl ethyl ketone to 2-methyl propanal decreased from 15.6 to 2.5. The configuration of 2,3-butanediol would affect the conversion but not the selectivity. The characterization results demonstrated that the phosphate modification of HZSM-5 could not only reduce the strong and medium acid sites but also produce new weak acid sites. Strong acid sites and high reaction temperatures could promote the formation of 2-methyl propanal through methyl migration via carboniums. Based on these results, a possible surface reaction model was proposed.

 Please wait while we load your content...
Please wait while we load your content...