N1-(3-Trimethoxysilylpropyl)diethylenetriamine grafted KIT-6 for CO2/N2 selective separation†
Abstract
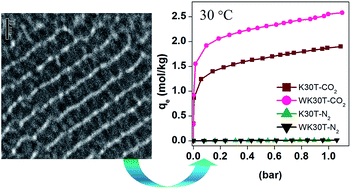
In the present study N1-(3-trimethoxysilylpropyl)diethylenetriamine was grafted on various ordered and commonly used mesoporous silica namely MCM-41 (2.2 nm), SBA-15 (6.6 nm) and KIT-6 (6.6 nm) in both anhydrous and aqueous conditions for CO2/N2 adsorption. The structural and physical properties before and after grafting were analyzed by nitrogen adsorption/desorption, X-ray diffraction and electron microscopy techniques. The uptake capacities of three-dimensional K30T and WK30T were 1.89 and 2.59 mol CO2 per kg of adsorbent, respectively at 30 °C which are significantly higher than MCM-41 and SBA-15 based adsorbents. Analysis of the enthalpy of CO2 adsorption, confirmed the adsorption in amine functionalized adsorbents by both chemical and physical interactions. Outstanding equilibrium CO2/N2 selectivity of functionalized KIT-6 over MCM-41 and SBA-15 opens up its practical applicability through a stable performance in several adsorptions/desorption cycles.

 Please wait while we load your content...
Please wait while we load your content...