Nose to brain delivery of astaxanthin-loaded solid lipid nanoparticles: fabrication, radio labeling, optimization and biological studies†
Abstract
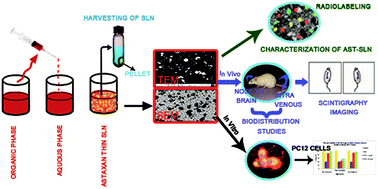
The present study was carried out to investigate the intranasal delivery of astaxanthin as solid lipid nanoparticles with the intention of improving brain targeting of astaxanthin for neurological disorders. The astaxanthin solid lipid nanoparticles were prepared by a double emulsion solvent displacement method. Statistical analysis using response surface methodology showed that the optimum values of stearic acid (50 mg), drug weight percentage (6.11%) and ratio of surfactant to co-surfactant (poloxamer 188 : lecithin-1 : 6) resulted in a 213.23 nm particle size and 0.367 polydispersity index for the astaxanthin solid lipid nanoparticles. Radio labeling studies were performed using technetium-99m to evaluate the biodistribution pattern after administration through different routes in experimental subjects. Radiolabeled nanoparticles were found to be 96–98% stable even after 48 h of labeling in phosphate-buffered saline (pH 7.4). Comparative biodistribution data indicated that the higher drug concentration in the brain was achieved by intranasal administration of 99mTc labeled astaxanthin solid lipid nanoparticles as compared to the intravenous route, which was also confirmed by gamma scintigraphy analysis. Furthermore, studies on the pheochromocytoma-12 cell line demonstrated the antioxidant potential of astaxanthin solid lipid nanoparticles against H2O2 induced toxicity. Our findings strongly emphasize that nanoparticle-based nasal drug delivery of astaxanthin could impart the utmost neuroprotection from oxidative stress in neurological disorders under in vitro conditions.

 Please wait while we load your content...
Please wait while we load your content...