A new approach toward the synthesis of 2,4-bis(fluoroalkyl)-substituted quinoline derivatives using fluoroalkyl amino reagent chemistry†
Abstract
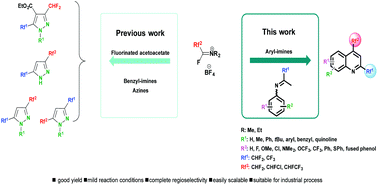
The present work describes the unprecedented use of Fluoroalkyl Amino Reagents (FARs) to afford 2,4-bis(fluoroalkyl)-substituted quinoline derivatives in two steps. In contrast to the Combes reaction, this approach allows for the synthesis of numerous quinoline derivatives bearing two identical or different fluoroalkyl substituents in the 2 and 4 positions, under mild reaction conditions, in good yields and with a very good regioselectivity. This reaction is easily scalable and suitable for an industrial process.
- This article is part of the themed collections: HOT articles in Organic Chemistry Frontiers in 2016 and Celebrating the 75th Birthday of Professor Barry Trost


 Please wait while we load your content...
Please wait while we load your content...