Homochiral association of binuclear trans-bis(β-iminoaryloxy)palladium(ii) complexes doubly linked with m-xylylene spacers: drastic linker-dependence of the association chirality of chiral clothespin-shaped molecules†
Abstract
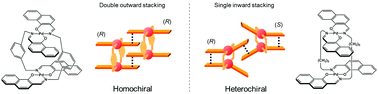
A significant effect of the linker on association chirality has been observed during the interpenetrative dimeric association of clothespin-shaped Pd complexes bearing either rigid or flexible linkers. Optically pure, planar, chiral binuclear trans-bis(β-iminoaryloxy)palladium(II) complexes doubly linked with m-xylylene spacers (1 and 2) were newly prepared by the reaction of Pd salts with the corresponding spacer-linked diimine ligands, and their rigid H-shaped conformation was confirmed by single-crystal XRD analysis. Analysis of the concentration dependence of optically pure and racemic mixtures of 1 in CDCl3 based on 1H NMR revealed that this complex exhibits homochiral association with an equilibrium constant ratio, [KD(homo)/KD(hetero)], of 1.2 × 102. This result is completely opposite to the reported heterochiral association of the pentamethylene-linked analogue 3, which has a KD(homo)/KD(hetero) value of 0.033 under the same conditions. Based on XRD analysis and molecular modelling, the drastic linker-dependence of the association chirality of 1 and 3 has been rationalized by proposing contrasting interpenetrative stacking modes during the dimeric association of these clothespin-shaped molecules. An outward double stacking mode, leading to homochiral association, was determined to be well-suited to the H-shaped, constrained conformation of the rigidly-linked 1, while an inward single stacking mode providing heterochiral association is preferred based on the A-shaped tilted conformation of the flexibly-linked 3.
- This article is part of the themed collection: Celebrating the 75th Birthday of Professor Barry Trost

 Please wait while we load your content...
Please wait while we load your content...