Redox-responsive, core-crosslinked degradable micelles for controlled drug release†
Abstract
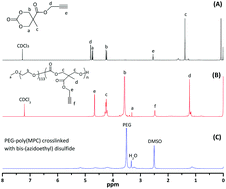
We developed novel redox-responsive, core-crosslinked micelles (CCLMs) via a simple, one-step click chemistry reaction. CCLMs were prepared by the click reaction between poly(ethylene glycol)-b-poly(5-methyl-5-propargylxycarbonyl-1,3-dioxane-2-one) [PEG-b-poly(MPC)] amphiphilic block copolymer and bis-(azidoethyl) disulfide. The CCLMs not only presented excellent stability under physiological conditions but also achieved the controlled release of DOX in reducing environment (such as DTT, GSH). Furthermore, DOX-loaded CCLMs showed low cytotoxicity in HeLa cells and 4T1 cells by MTT assay as well as enhanced cytotoxicity against drug-resistant ADR/MCF-7 cells in vitro. Confocal laser scanning microscope (CLSM) images show that the DOX-loaded materials were easily taken up by HeLa cells, compared to free DOX as control. With its facile preparation, superior stability, and controlled release, the redox-responsive CCLMs can provide a versatile platform for drug delivery and have great potential for antitumor therapy.

 Please wait while we load your content...
Please wait while we load your content...