A nitrogen-rich, azaindole-based microporous organic network: synergistic effect of local dipole–π and dipole–quadrupole interactions on carbon dioxide uptake†
Abstract
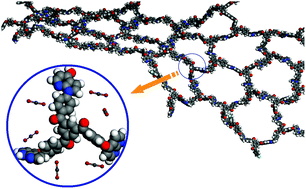
A new type of microporous organic polymer with azaindole units (N-PEINK) has been designed. The resulting N-PEINK exhibits good chemical and thermal stability with a decomposition temperature of 550 °C. Taking advantage of the synergistic effect of local dipole–π and dipole–quadrupole interactions between azaindole and carbon dioxide (CO2), the CO2 uptake capacity of the polymer reaches 20.8 wt% (1.0 bar, 273 K) with high selectivities (CO2/N2 = 97, CO2/CH4 = 18), making the polymer a promising microporous material for application in CO2 separation and capture. Furthermore, the azaindole-based microporous organic polymer also exhibits a high hydrogen storage (2.67 wt%) at 1.0 bar and 77 K. For comparison, the microporous organic polymer with indole units (PEINK) was also prepared.

 Please wait while we load your content...
Please wait while we load your content...