Tough and degradable photopolymers derived from alkyne monomers for 3D printing of biomedical materials†
Abstract
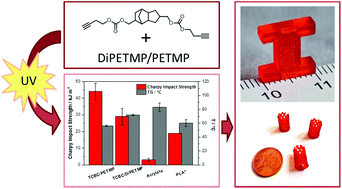
This contribution deals with the synthesis and exploration of alkyne carbonate derivatives as biocompatible building blocks in the thiol–yne photopolymerisation reaction with the aim to facilitate the fabrication of tailor made medical devices by UV based additive manufacturing technologies. It turned out that the investigated alkyne carbonates offer curing rates similar to comparable acrylates, while providing much higher conversion and lower monomer cytotoxicity. Curing the synthesized building blocks in combination with the commercially available thiol pentaerythritol tetra(3-mercaptopropionate) (PETMP) leads to networks that degrade in aqueous alkaline and acidic media in a surface erosion manner. Additionally, a selective adjustment of the degradability is feasible by the choice and content of thiol monomers. Notably, monomers containing a tricyclo[5.2.1.02,6]decane-4,8-dimethanol backbone provide decent thermo-mechanical properties and appropriate impact strengths similar to polylactic acid (PLA). Most importantly, selected thiol–yne formulations were printed successfully with an accuracy of 40 × 40 μm, which seems to be sufficiently high to prints medical devices in appropriate resolution.

 Please wait while we load your content...
Please wait while we load your content...