Temperature and pH dual-responsive poly(vinyl lactam) copolymers functionalized with amine side groups via RAFT polymerization†
Abstract
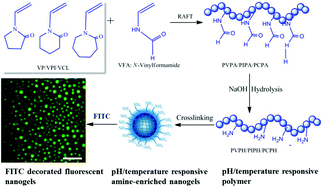
A series of well-defined statistical copolymers based on cyclic N-vinyl lactams (N-vinylcaprolactam, N-vinylpiperidine and N-vinylpyrrolidone) and N-vinylformamide (VFA) were successfully synthesized via RAFT polymerization. The less-polar solvents (anisole and 1,4-dioxane) were more favorable over the highly-polar solvents (DMSO and DMF) while 2-(ethoxycarbonothioylthio)propanoate was more suitable than dithiobenzoate derivatives as the chain transfer agent (CTA) for copolymerization. The optimal polymerization conditions were explored with methyl 2-(ethoxycarbonothioylthio)propanoate as the CTA and anisole as the solvent. The narrow dispersity (Đ) of the copolymers, linear relationship between conversion and molecular weight, and symmetrical unimodal GPC traces confirmed the controlled character of the polymerization process. The polymerization process was progressively disfavored by the enhanced steric hindrance from the enlarged lactam ring while the VFA composition could be well controlled up to as high as 50% by continuously feeding VFA with a syringe pump. The lower critical solution temperatures (LCSTs) of the thermo-responsive copolymers poly(N-vinylcaprolactam-co-N-vinylformamide) and poly(N-vinylpiperidine-co-N-vinylformamide) in aqueous solutions reflected by the appearance of the cloud points can be tunable by the molecular weights and compositions of the copolymers. Primary amine functionalized temperature/pH dual responsive reactive copolymers were obtained by the controlled hydrolysis of the VFA components under alkaline conditions. The influence of the lactam ring on the hydrolysis rates was investigated. The LCSTs of the hydrolyzed copolymers can be elegantly adjusted by the hydrolysis degree. Amine-enriched nanogels were prepared in W/O emulsions utilizing the coupling between the reactive amine side-groups of polymer chains and diacrylate functionalities of a poly(ethylene glycol) cross-linker via Michael addition reaction, which displayed temperature and pH double responsiveness. The nanogels were labelled with fluorescein isothiocyanate (FITC) through conjugation with the residual primary amine groups on the surface. This facile chemistry has opened up routes to synthesize water-soluble reactive stimuli-responsive copolymers with well-defined architectures for design of functional macromolecular materials.

 Please wait while we load your content...
Please wait while we load your content...