Complexation of thermoresponsive dialkoxynaphthalene end-functionalized poly(oligoethylene glycol acrylate)s with CBPQT4+ in water†
Abstract
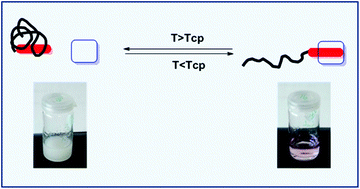
Four different oligoethylene glycol acrylates (OEGA), namely hydroxypropylacrylate (HPA), methoxy diethylene glycol acrylate (mDEGA), methoxy triethylene glycol acrylate (mTEGA) and 2-hydroxyethylacrylate (HEA) were homopolymerized via RAFT polymerization employing a naphthalene functionalized chain transfer agent resulting in thermoresponsive naphthalene-functionalized POEGAs with different hydrophilicities. Supramolecular inclusion complexes of these POEGAs with electron-deficient cyclophane cyclobis(paraquat-p-phenylene) tetrachloride (CBPQT4+) in water were studied. The association constants (Ka) were determined to study the effect that polymer hydrophilicity has on the Ka and results indicated that the nature of the polymer did not significantly influence the complexation strength and the association is mostly enthalpy driven. The impact of temperature on the host–guest complexes was also investigated. A continuous partial thermally induced dissociation of complexes was observed upon raising the temperature with a more distinct decrease in complexation around the cloud point temperature (TCP) of the POEGA employed, indicating the importance of the polymer phase transition for tuning the recognition properties of dialkoxynaphthalene end-decorated poly(oligoethylene glycol acrylate)s in water.

 Please wait while we load your content...
Please wait while we load your content...