Synthesis and properties of an acid-labile dual-sensitive ABCD star quaterpolymer†
Abstract
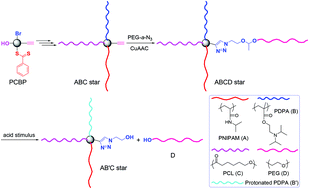
Synthesis of multicomponent miktoarm stars (MMS) has attracted much attention due to their intriguing morphologies and multipurpose applications. Thus far, the examples of stimuli-cleavable MMS are very scarce. This study aims at modular synthesis of the first example of an acid-labile ABCD star quaterpolymer and exploring its potential in smart drug delivery systems. An alkyne-core-functionalized ABC star terpolymer was initially synthesized via the “core-first” method, and then CuAAC was performed to achieve the desired star with poly(N-isopropylacrylamide) (PNIPAM, A), poly(2-diisopropylaminoethyl methacrylate) (PDPA, B), poly(ε-caprolactone) (PCL, C), and acetal-linked poly(ethylene glycol) (aPEG, D) segments. Upon an acid stimulus, the ABCD star was liable to “degrade” into a mixture of PEG and AB′C star with a protonated PDPA (B′) segment, and the differences in the topology and composition further induced time-dependent morphological transformations of copolymer aggregates. With prolonging time, various morphologies involving large compound micelles (0 h), flower-like micelles (1 h), small micelles and their aggregates (3 h), and compound micelles with a reduced size (t ≥ 12 h) were observed from TEM images. Doxorubicin-loaded copolymer aggregates exhibited accelerated drug release kinetics upon thermo and pH stimuli, and the micellar system may hold great promise for biomedical applications due to its relatively low cytotoxicity and stimuli-tunable release properties. In addition to developing a modular “3 + 1” approach to generate star quaterpolymers, this study underlies systematic research studies on properties and applications of functional MMS and their derivatives.

 Please wait while we load your content...
Please wait while we load your content...