Bio-based difuranic polyol monomers and their derived linear and cross-linked polyurethanes†
Abstract
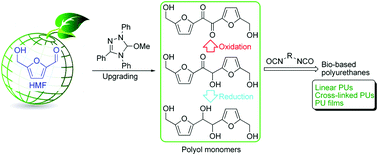
A triol monomer, 5,5′-dihydroxymethyl furoin (DHMF), was prepared from the biomass platform chemical 5-hydroxymethylfurfural (HMF) in 95% yield via organocatalysis. Selective oxidation and reduction of DHMF afforded a new diol monomer, 5,5′-bihydroxymethyl furil (BHMF), and a new tetraol, 5,5′-bihydroxymethyl hydrofuroin (BHMH), respectively. The catalyzed polyaddition of the diol BHMF with various diisocyanates produced linear polyurethanes (PUs), whereas catalyzed polyadditions using the triol and tetraol monomers led to cross-linked PUs. Especially interesting is the PU material derived from BHMF and aromatic diisocyanates such as diphenylmethane diisocyanate, which exhibited a Mn of 39.8 kg mol−1, an onset decomposition temperature of 234 °C, and a Tg of 140 °C. Various PU thin films have also been prepared by in situ polyaddition of these three polyols with diisocyanates in various ratios through solvent casting, affording PU materials ranging from being brittle to flexible with a high strain at break of 300%.

 Please wait while we load your content...
Please wait while we load your content...