Oxidation and temperature dual responsive polymers based on phenylboronic acid and N-isopropylacrylamide motifs†
Abstract
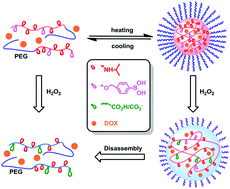
Oxidation stress has been becoming an important target for the development of smart nanomedicines, triggering research interest in oxidation responsive polymers. Herein, we report a new type of temperature/oxidation dual responsive copolymer. They were synthesized by the sequential atom transfer radical copolymerization (ATRP) of N-isopropylacrylamide (NIPAM, M1) and a phenylboronic pinacol ester-containing acrylate (M2) and dialysis against water to remove the pinacol protecting groups. The copolymers with a small amount of phenylboronic acid units (<7%) were soluble in cold neutral phosphate buffer but showed lower critical solution temperatures in the range of 12–31 °C. The thermally induced phase transition profiles depended on both the composition and concentration of the polymers. The cloud points of the copolymers were shifted to higher temperatures upon H2O2 induced oxidation of the phenylboronic acid and the subsequent 1,6-elimination. Using PEG-Cl as the macroinitiator to initiate ATRP of M1 and M2, three block copolymers composed of a PEG block and a temperature/oxidation dual responsive segment were prepared after dialysis in water. These block copolymers had a similar ratio of NIPAM to phenylboronic acid units but different molecular weights. We have studied their thermal self-assembly and H2O2 triggered decomposition by laser light scattering, 1H NMR, and transmission electron microscopy. Upon a fast heating protocol, these block copolymers formed stable micelle-like nanoparticles (at 37 °C) that were capable of encapsulating doxorubicin (DOX) and showed H2O2 triggered release. The naked nanoparticles were cytocompatible, however the DOX-loaded ones exhibited concentration dependent cytotoxicity, in particular to cancer cells.

 Please wait while we load your content...
Please wait while we load your content...