An ultra-narrow bandgap derived from thienoisoindigo polymers: structural influence on reducing the bandgap and self-organization†
Abstract
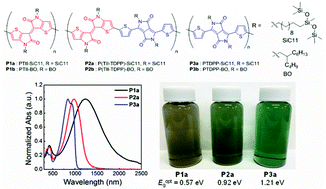
Six conjugated polymers based on thienoisoindigo (TII) and thiophene-flanked diketopyrrolopyrrole (TDPP) units bearing either branched-alkyl or siloxane-terminated alkyl solubilizing groups have been synthesized. We report the impact of backbone selection and side chain engineering on the optical, electrochemical, and solid state packing, as well as carrier transport properties, for this set of polymers. The optical bandgap of the polymers is shown to systematically decrease on TII unit incorporation into the polymer chain. TII homopolymers absorb near infrared wavelengths and have extremely narrow optical band gaps below 0.60 eV, mainly as a result of the contribution of the flat and rigid π-frameworks corroborated with a strong quinoidal character of the TII unit. The theoretical calculations and Raman spectra measurements well demonstrate that the presence of a TII unit contributes to π-delocalization inducted by quinoidal resonance. Our X-ray analyses indicate that the TII unit prefers face-on lamellar orientations compared with the TDPP unit, and the largely separated branching point over the undecyl spacer of siloxane-terminated chains reinforces π–π interchain interactions in a crystalline film.


 Please wait while we load your content...
Please wait while we load your content...