A systematic study of protein labeling by fluorogenic probes using cysteine targeting vinyl sulfone-cyclooctyne tags†
Abstract
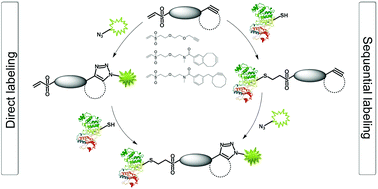
Fluorescent tagging of proteins via accessible cysteine residues is of paramount importance. In this study, model proteins of interest (mitogen-activated protein kinases) were labeled successfully in native state on their free thiols by direct fluorescence derivatization, or in a sequential manner where conjugation of the site specific linker and the fluorophore is carried out in two steps. To this end we designed and prepared two novel chemical reporters carrying vinyl sulfone as Cys targeting function and cyclooctyne motifs, suitable for subsequent conjugation with fluorogenic azides via copper free strain-promoted azide–alkyne click chemistry. Direct and sequential labeling reaction steps were analyzed by native PAGE, capillary zone electrophoresis and tandem mass spectrometry. The efficiency of tagging was correlated with solvent accessibility of the Cys residues. Our results indicated that conjugation of native proteins by vinyl sulfone linkers was fast and thiol-selective. Subsequent click reaction with fluorogenic dyes generates intensive fluorescence signals and fulfills all requirements of bioorthogonality.

 Please wait while we load your content...
Please wait while we load your content...