A FeCl3-catalyzed highly regioselective 1,2-addition/substitution sequence for the construction of coumarin-substituted bis(indolyl)methanes†
Abstract
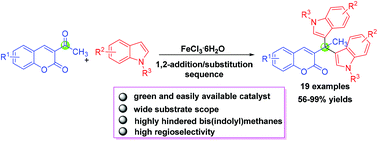
A FeCl3-catalyzed highly regioselective 1,2-addition/substitution sequence of 3-acetylcoumarins and indoles has been developed to afford highly hindered tetrasubstituted bis(indolyl)methanes bearing a biologically useful coumarin motif in 56–99% yields.

 Please wait while we load your content...
Please wait while we load your content...