A convenient domino Ferrier rearrangement-intramolecular cyclization for the synthesis of novel benzopyran-fused pyranoquinolines†
Abstract
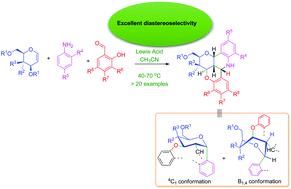
The Ferrier rearrangement and the Povarov reaction have proven indispensable tools in carbohydrate chemistry and the synthesis of N-heterocycles, respectively. We hereby report a one-pot cyclization sequence involving the Ferrier and Povarov-like reactions in the synthesis of novel pentacyclic N-heterocycles: benzopyran-fused pyranoquinolines. The reaction entails three component condensation of a glycal with a variety of anilines and 2-hydroxybenzaldehydes under Lewis acid catalysis to yield the title compounds in 4–24 hours of reaction time, in moderate to high yields and excellent diastereoselectivity. Of the Lewis acid catalysts deployed [Sc(OTf)3, Al(OTf)3, Cu(OTf)2, CuOTf, I2, InCl3, and La(OTf)3] in various solvents (acetonitrile, THF, dichloromethane, 1,2-dichloroethane and diethyl ether) at room and elevated temperatures, Sc(OTf)3 (10 mol%) in acetonitrile at 70 °C gave the best results, with excellent diastereoselectivity. CAN-mediated oxidative ring opening of the pentacyclic N-heterocycle gave the corresponding enantiometrically pure chromenoquinoline bearing a pendant sugar moiety.
- This article is part of the themed collection: New Talent

 Please wait while we load your content...
Please wait while we load your content...