In situ formation of a ZnO/ZnSe nanonail array as a photoelectrode for enhanced photoelectrochemical water oxidation performance†
Abstract
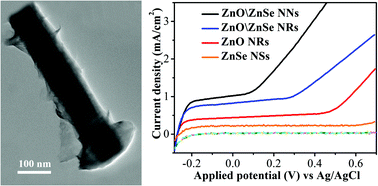
In this study, a ZnO/ZnSe nanonail array was prepared via a two-step sequential hydrothermal synthetic route. In this synthetic process, the ZnO nanorod array was first grown on a fluorine-doped tin oxide (FTO) substrate using a seed-mediated growth approach via the hydrothermal process. Then, the ZnO nanonail array was obtained via in situ growth of ZnSe nano caps onto the ZnO nanorod array via a hydrothermal process in the presence of a Se source. The surface morphology and amount of ZnSe grown on the surface of the ZnO nanorods can be regulated by varying the reaction time and reactant concentration. Compared with pure ZnO nanorods, this unique nanonail array heterostructure exhibits enhanced visible light absorption. The transient photocurrent condition, in combination with steady-state and time-resolved photoluminescence spectroscopy, reveals that the ZnO/ZnSe nanonail array electrode has the highest charge separation rate, highest electron injection efficiency, and highest chemical stability. The photocurrent density of the ZnO/ZnSe nanonail array heterostructure reaches 1.01 mA cm−2 at an applied potential of 0.1 V (vs. Ag/AgCl), which is much higher than that of the ZnO/ZnSe nanorod array (0.71 mA cm−2), the pristine ZnO nanorod array (0.39 mA cm−2), and the ZnSe electrode (0.21 mA cm−2), indicating its significant visible light driven activities for photoelectrochemical water oxidation. This unique morphology of nail-capped nanorods might be important for providing better insight into the correlation between heterostructure and photoelectrochemical activity.

 Please wait while we load your content...
Please wait while we load your content...