Strong metal–support interaction in novel core–shell Au–CeO2 nanostructures induced by different pretreatment atmospheres and its influence on CO oxidation†
Abstract
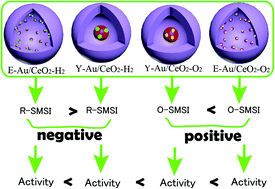
Yolk–shell Au/CeO2 (Y-Au/CeO2) and encapsulated Au/CeO2 (E-Au/CeO2) nanocatalysts were prepared by using silica templates. A strong metal–support interaction (SMSI) in the Au/CeO2 nanostructures induced by different pretreatment atmospheres and its influence on CO oxidation were studied. E-Au/CeO2 pretreated in O2 had the best performance, followed by Y-Au/CeO2 pretreated in O2, Y-Au/CeO2 pretreated in H2, and E-Au/CeO2 pretreated in H2. The reasons for the different activities were discussed. There were two kinds of strong metal–support interactions (SMSI) between Au and CeO2 termed as R-SMSI (pretreated in reductive atmosphere) and O-SMSI (pretreated in oxidation atmosphere). Because of the smaller size of the Au and the larger contact area, both the R-SMSI and O-SMSI of E-Au/CeO2 were larger than those of Y-Au/CeO2. The O-SMSI was accompanied by the formation of cationic Au species that were beneficial to the enhancing of activity. As expected, the activity of E-Au/CeO2 pretreated in O2 with a Au size less than 5 nm was higher than that of Y-Au/CeO2 pretreated in O2 with 25 nm Au. However, it is surprisingly found that the activity of Y-Au/CeO2 pretreated in H2 with 25 nm Au was higher than that of E-Au/CeO2 pretreated in H2 with a Au size less than 5 nm. R-SMSI resulted in the formation of a AuCe alloy that had a negative effect on the activity. Compared with E-Au/CeO2 pretreated in H2, Y-Au/CeO2 pretreated in H2 exhibited a smaller relative content of the AuCe alloy, leading to a better activity of Y-Au/CeO2 pretreated in H2.

 Please wait while we load your content...
Please wait while we load your content...