3-Acylated tetramic and tetronic acids as natural metal binders: myth or reality?†
Abstract
Covering: up to 2015
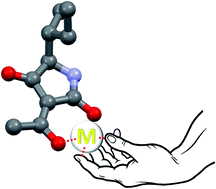
3-Acylated tetramic and tetronic acids are characterized by a low pKa and are likely to be deprotonated under physiological conditions. In addition, their structure makes them excellent chelators of metallic cations. We will discuss the significance of these chemical properties with regard to the biological properties and mechanisms of action of these compounds, highlighting the importance of considering them as salts or chelates for biological purposes, rather than acids.

 Please wait while we load your content...
Please wait while we load your content...