Photodegradable self-assembling PAMAM dendrons for gene delivery involving dendriplex formation and phototriggered circular DNA release†
Abstract
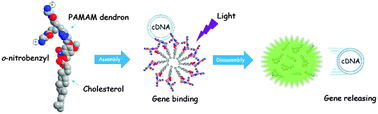
For effective gene delivery, structural degradation of synthetic carriers is crucial to the release of nucleic acids on the transfection time scale. In this study, we have synthesized the amphiphilic dendritic scaffolds with a photolabile o-nitrobenzyl (o-NB) group that can enable the structural decomposition and controlled release of nucleic acids under active light stimulation. The amphiphilic counterpart composed of a lipophilic cholesterol and a hydrophilic poly(amido amine) (PAMAM) dendron allows the self-assembly into a core–shell-like pseudodendrimer above the critical aggregation concentration (CAC) of approximately 20 μM. On the basis of electrostatic interaction, the polycationic pseudodendrimers are capable of forming stable complexes with polyanionic cyclic reporter genes under low charge excess values, suggesting substantial binding affinity of the dendron assembly toward circular DNA. Because the o-NB group in the dendritic structure undergoes efficient photolytic cleavage, an in vitro test shows that thus-formed “dendriplexes” are readily dissociated under 365 nm light irradiation, causing effective dendron degradation accompanied by DNA release. This photochemical strategy provides an opportunity to control gene binding and release in a spatiotemporal manner.

 Please wait while we load your content...
Please wait while we load your content...