Mono and trivalent thallium–sulfur interactions and their influence on the formation of nano thallium sulphide: single crystal X-ray structural and spectral studies on thallium(i)/(iii)–cyclohexylpiperazine dithiocarbamates†
Abstract
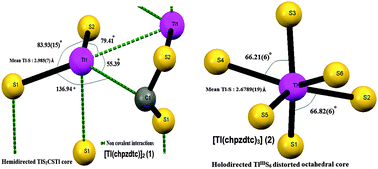
Thallium(I) and thallium(III)–cyclohexylpiperazine dithiocarbamates have been prepared with the same donor environment for the first time and analyzed by electronic, IR, 1H, 13C NMR spectral, CV and single crystal X-ray structural analyses. Solvothermal decomposition of the dithiocarbamates yielded nano Tl4S3 and were characterized. Bond parameters from single crystal X-ray structures have been used in continuous symmetry measure and bond valence sum analysis of the compounds to establish the octahedral geometry and formal oxidation numbers of thallium. [Tl(chpzdtc)]2 (1) shows extensive non covalent interactions and the hemidirected TlS2CSTl core is of distorted square pyramidal geometry with the stereo chemically active lone pair of thallium(I) occupying a vertex of the square pyramid. This is the first report which identifies the 4f7/2 and 4f5/2 electron binding energies of Tl(I) and Tl(III) dithiocarbamates unambiguously and the effect of Tl⋯C interaction on XPS binding energies. [Tl(chpzdtc)3] (2), the trivalent analogue as a contrast is a typical holodirected TlS6 distorted octahedral core with no significant supramolecular interactions. Mean Tl–S bond distances in 1 and 2 are 2.985(7) and 2.6789(19) Å, respectively, which clearly support the higher ease of solvothermal decomposition of 1 to nano Tl4S3 than 2. A strong correlation exists between the bond strengths of Tl–S, the thioureide C–N and the ease of formation of Tl4S3 from the two dithiocarbamates 1 and 2.

 Please wait while we load your content...
Please wait while we load your content...