Synthesis and conformation of a novel fluorescein–Zn-porphyrin dyad and intramolecular energy transfer†
Abstract
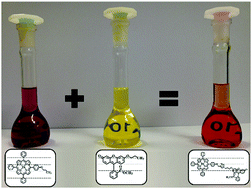
This study describes the synthesis and characterization of a new zinc porphyrin–fluorescein dyad, the two chromophoric units being covalently linked by a 1,2,3-triazole bridge. The latter was formed by Cu-catalyzed Huisgen 1,3-dipolar cycloaddition. The conformational analysis of this dyad (1) was performed by NOESY experiments, suggesting interactions between moieties; density functional theory (DFT) calculations confirmed clear evidence of a folded conformer, which is stabilized by electrostatic and CH–π interactions. Photophysical measurements demonstrated solvent-dependent energy transfer, with an efficiency of about 40%.

 Please wait while we load your content...
Please wait while we load your content...