Multiple di-leucines in the ATP7A copper transporter are required for retrograde trafficking to the trans-Golgi network†
Abstract
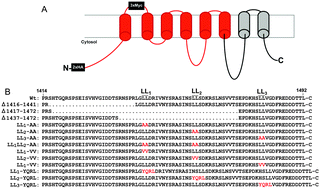
The ATP7A protein is a ubiquitous copper-transporting P-type ATPase that is mutated in the lethal pediatric disorder of copper metabolism, Menkes disease. The steady-state location of ATP7A is within the trans-Golgi network (TGN), where it delivers copper to copper-dependent enzymes within the secretory pathway. However, ATP7A constantly cycles between the TGN and the plasma membrane, and in the presence of high copper concentrations, the exocytic arm of this cycling pathway is enhanced to promote a steady-state distribution of ATP7A to post-Golgi vesicles and the plasma membrane. A single di-leucine endocytic motif within the cytosolic carboxy tail of ATP7A (1487LL) was previously shown to be essential for TGN localization by functioning in retrieval from the plasma membrane, however, the requirement of other di-leucine signals in this region has not been fully investigated. While there has been some success in identifying sequence elements within ATP7A required for trafficking and catalysis, progress has been hampered by the instability of the ATP7A cDNA in high-copy plasmids during replication in Escherichia coli. In this study, we find that the use of DNA synthesis to generate silent mutations across the majority of both mouse and human ATP7A open reading frames was sufficient to stabilize these genes in high-copy plasmids, thus permitting the generation of full-length expression constructs. Using the stabilized mouse Atp7a construct, we identify a second di-leucine motif in the carboxy tail of ATP7A (1459LL) as essential for steady-state localization in the TGN by functioning in endosome-to-TGN trafficking. Taken together, these findings demonstrate that multiple di-leucine signals are required for recycling ATP7A from the plasma membrane to the TGN and illustrate the utility of large-scale codon reassignment as a simple and effective approach to circumvent cDNA instability in high-copy plasmids.
- This article is part of the themed collection: Mammalian Copper Transport and Related Disorders

 Please wait while we load your content...
Please wait while we load your content...