FecB, a periplasmic ferric-citrate transporter from E. coli, can bind different forms of ferric-citrate as well as a wide variety of metal-free and metal-loaded tricarboxylic acids†
Abstract
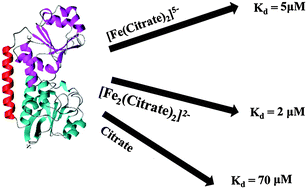
The Escherichia coli Fec system, consisting of an outer membrane receptor (FecA), a periplasmic substrate binding protein (FecB) and an inner membrane permease-ATPase type transporter (FecC/D), plays an important role in the uptake and transport of Fe3+-citrate. Although several FecB sequences from various organisms have been reported, there are no biophysical or structural data available for this protein to date. In this work, using isothermal titration calorimetry (ITC), we report for the first time the ability of FecB to bind different species of Fe3+-citrate as well as other citrate complexes with trivalent (Ga3+, Al3+, Sc3+ and In3+) and a representative divalent metal ion (Mg2+) with low μM affinity. Interestingly, ITC experiments with various iron-free di- and tricarboxylic acids show that FecB can bind tricarboxylates with μM affinity but not biologically relevant dicarboxylates. The ability of FecB to bind with metal-free citrate is also observed in 1H,15N HSQC-NMR titration experiments reported here at two different pH values. Further, differential scanning calorimetry (DSC) experiments indicate that the ligand-bound form of FecB has greater thermal stability than ligand-free FecB under all pH and ligand conditions tested, which is consistent with the idea of domain closure subsequent to ligand binding for this type of periplasmic binding proteins.

 Please wait while we load your content...
Please wait while we load your content...