Deferasirox-coated iron oxide nanoparticles as a potential cytotoxic agent†
Abstract
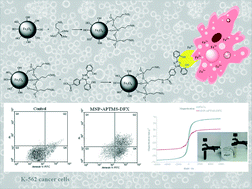
Two broad strategies for the use of iron chelators in cancer treatment have been explored. The first was using iron chelators to reduce the iron level in cancer cells. Cancer cells typically require more iron than normal cells to mediate their generally rapid DNA synthesis and growth. The second more recent strategy is using chelators that help the redox cycling of iron to generate cytotoxic ROS (reactive oxygen species) within tumors. Both methods are currently being pursued. Deferasirox is an FDA-approved medication used to reduce chronic iron overload. Because of the low toxicity and oral administration of deferasirox, the use of this drug in cancer therapy is a new approach. Therefore, a number of recent studies have examined the potential of deferasirox as an anticancer agent. According to these strategies, the present study has reported the new role of deferasirox (DFX) loaded on iron oxide nanoparticles (Fe3O4@DFX) as an anticancer agent. The cytotoxicity of Fe3O4@DFX was screened against MCF-7, HeLa, HT-29, K-562, Neuro-2a, and L-929 cell lines by MTT assay. The surface of magnetic nanoparticles (MNPs) was first coated with (3-aminopropyl) trimethoxysilane (APTMS) and was then linked with deferasirox via an amidation reaction between –NH2 and –COOH to form well-dispersed surface-functionalized biocompatible MNPs. The obtained nanoparticles were thoroughly characterized by various spectroscopic and microscopic methods such as SEM, FT-IR, TGA and VSM. Because of the ability of magnetic nanoparticles in biomedical applications to be targeted toward the site of action using magnetic field gradients, we suggest that MNPs loaded with deferasirox could be considered as a potential formulation, which can simultaneously be used as a theranostic agent for both cancer treatment and imaging. The obtained results provide experimental evidence that magnetite nanoparticles loaded with deferasirox induce apoptosis in cancer cell lines. Moreover, flow cytometry results confirm that the investigated compound produces a high population of apoptotic cells (69.3%), 1.2 fold higher than cisplatin (58.1%) at the same concentration, and could induce apoptosis in human leukemia cell lines (K-562).

 Please wait while we load your content...
Please wait while we load your content...