Tabtoxinine-β-lactam is a “stealth” β-lactam antibiotic that evades β-lactamase-mediated antibiotic resistance†‡§
Abstract
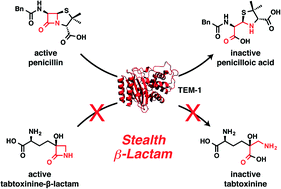
Tabtoxinine-β-lactam (TβL) is a phytotoxin produced by plant pathogenic strains of Pseudomonas syringae. Unlike the majority of β-lactam antibiotics, TβL does not inhibit transpeptidase enzymes but instead is a potent, time-dependent inactivator of glutamine synthetase, an attractive and underexploited antibiotic target. TβL is produced by P. syringae in the form of a threonine dipeptide prodrug, tabtoxin (TβL-Thr), which enters plant and bacterial cells through dipeptide permeases. The role of β-lactamases in the self-protection of P. syringae from tabtoxin has been proposed, since this organism produces at least three β-lactamases. However, using in vitro and cellular assays and computational docking we have shown that TβL and TβL-Thr evade the action of all major classes of β-lactamase enzymes, thus overcoming the primary mechanism of resistance observed for traditional β-lactam antibiotics. TβL is a “stealth” β-lactam antibiotic and dipeptide prodrugs such as tabtoxin from P. syringae represent a novel antibiotic therapeutic strategy for treating multi-drug resistant Gram-negative pathogens expressing high levels of β-lactamase enzymes.

 Please wait while we load your content...
Please wait while we load your content...