Structure and dynamics of H. pylori 98-10 C5-cytosine specific DNA methyltransferase in complex with S-adenosyl-l-methionine and DNA†
Abstract
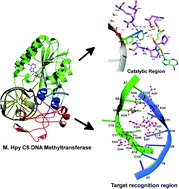
Helicobacter pylori is a Gram-negative bacterium that inhabits the human gastrointestinal tract, and some strains of this bacterium cause gastric ulcers and cancer. DNA methyltransferases (MTases) are promising drug targets for the treatment of cancer and other diseases that are also caused by epigenetic alternations of the genome. The C5-cytosine specific DNA methyltransferase from H. pylori (M. Hpy C5mC) catalyzes the transfer of the methyl group from the cofactor S-adenosyl-L-methionine (AdoMet) to the flipped cytosine of the substrate DNA. Herein we report the sequence analyses, 3-D structure modeling and molecular dynamics simulations of M. Hpy C5mC, when complexed with AdoMet as well as DNA. We analyzed the protein–DNA interactions prominently established by the flipped cytosine and the interactions between the protein and cofactor in the active site. We propose that the contacts made by cytosine O2 with Arg155 and Arg157, and the water-mediated interactions with cytosine N3 may be essential for the activity of methyl transfer as well as the deprotonation at the C5 position in our C5mC model. Specific recognition of DNA was mediated mainly by residues from Ser221–Arg229 and Ser243–Gln246 of the target recognition domain (TRD) and some residues of the loop Ser75–Lys83 from the large domain. These findings are further supported by alanine scanning mutagenesis studies. The results reported here explain the sequence, structure and binding features necessary for the recognition between the cofactor and the substrate by the key epigenetic enzyme, M. Hpy C5mC.

 Please wait while we load your content...
Please wait while we load your content...